
Water scarcity is a well-known problem that we are facing today. Our population is growing at an enormous rate, and pretty soon we will not have enough water to provide for everyone.
The sea covers almost 70% of the earth’s surface area which makes it a water source that will likely never run out.
There is only one problem.
Because of its high salt content, we cannot directly use it, and it needs to go through several expensive processes to be converted into fresh drinkable water.
Currently, several projects are being tested for its effectiveness, cost and also its effect on the environment.
I am going to cover some of the fundamental challenges that the process of desalination is facing. I am also going to cover up to 12 different desalination methods that are being tested and also being used today.
I am also going to show you how to turn seawater into fresh drinkable water if you should ever find yourself stranded somewhere along or on the sea.
The Challenges Of Desalination
Below, I will cover some of the fundamental challenges associated with the desalination of water. I will include a method that mother nature relies on and also some issues related to cost and the environment.
a) We Are Running Out Of Water
Desalination is the process where salt is being taken out of the water and turning it into fresh drinking water. On land our fresh water supply is limited, but we have an overabundance of water in the sea. The only problem is that we cannot use this water and it is quite a challenge to turn it into usable water.
Get this.
It is also said that less than 1% of the water on this planet is drinkable. Another problem to consider is that our global population is growing each year by at least 1 billion which mean that our demand for water is also growing.
Here is an example.
Before the 1800s we had less than a billion people living on the planet. We have grown to over 7 billion in more than 220 years later. This is the reason why we need to look at alternative water sources and why not the sea. We need to find a solution and fast.
b) We Can Learn A Lot From Mother Nature
Mother nature already has a process installed that turns salt water into fresh drinkable water. One method that nature uses is evaporation with a process called the hydrological cycle. This refers to the process where water is continuously moving below and above the surface of our planet.

The sun will evaporate the water from the sea leaving the salt behind because salt cannot evaporate. Freshwater will then fill the air in the form of vapor and will then form clouds in the sky. The clouds will then produce fresh water in the form of rain.
It is said that the sea covers 70% of our planets surface area. This totals 510 million square kilometers which means that our planet has a lot of water to work with. The soil and sand around the sea also act as a natural filter which is helpful at removing salt from the water.
c) It Is Not Cost-Efficient
It is a well-known fact that cleaning the salt from water is very expensive. Governments from around the world are working on projects to develop methods that are sustainable both financially and environmentally.
Many factors will influence the cost of this process. One factor that can attribute up to 50% of the price is electricity. Currently, it is much cheaper to directly extract fresh water from sources such as rivers and dams. Removing salt from the water can cost up to 10 times more than the previously mentioned sources.
It is getting cheaper because there is more supply to some of the materials needed to filter the water. There is also the cost of transport for if the desalination plants are inland instead of operating from the coast.
So how much does it cost?
It is well estimated that it can cost up to $2 to filter one cubic meter of salt water. If the water is sourced from rivers, it can cost around 15 cents which is much cheaper. The majority of these plants are located in the middle east where oil is cheap enough to drive these plants profitably.
As a result of these factors, the location of the desalination plant will contribute significantly to how much it will cost. We also need to consider other factors such as waste products and also extra filters to prevent fish asd plankton from entering the factories.
d) Is It Environmentally Friendly?
Desalination does have a negative impact on the environment directly and indirectly. The direct effects it has on the environment is the by-products. The question is, what happens to the excess salt that is separated from the water?
Some factories might sell some of it while others will dump it back into the sea. By dumping it into the sea, we will increase the salt content of the water which might have a negative influence on marine life.
In regards to the negative impact, we can refer to its carbon footprint created by its large electricity demand. We are still in the beginning phases of implementing this process, so the carbon footprint created is not that large.
The problem comes when we start to do it at scale. If we get to a point where most of our freshwater sources are gone, and we need to rely on cleaning out seawater, then the carbon footprint left by the factories will become significant.
Modern Applications
Currently, we already have several desalination plants up and running worldwide and we also make use of different methods that need to be tested and evaluated for its effectiveness.
a) Pervaporation
This method can also be called pervaporative separation that is an eco-friendly and inexpensive way to remove the salt from seawater. It is a 2-step process that filters the water with the help of a polymeric membrane which is the first step.

Some membranes can also be embedded by cellulose acetate powder which is an excellent attractant for salt and comes from wood pulp.
The second step takes the filtered water and then heat it until it evaporates. It is important to note that the second step does not necessarily need electricity to produce the heat as it can use the heat coming from the sun to evaporate the water.
The first step has already filtered out the larger particles and does not require that much energy for the vaporization stage. The second stage only allows for the removal of the smaller particles. The water is then condensed into drinkable and clean water.
Another reason why this method is very favorable is because the materials used can be found anywhere in the world which makes it cheap to manufacture.
b) Membrane distillation
This process involves the separation of salt and water which is driven by the change in temperature. This process does make use of electricity to generate heat but can be used with the help of solar panels which also contributes to green energy.
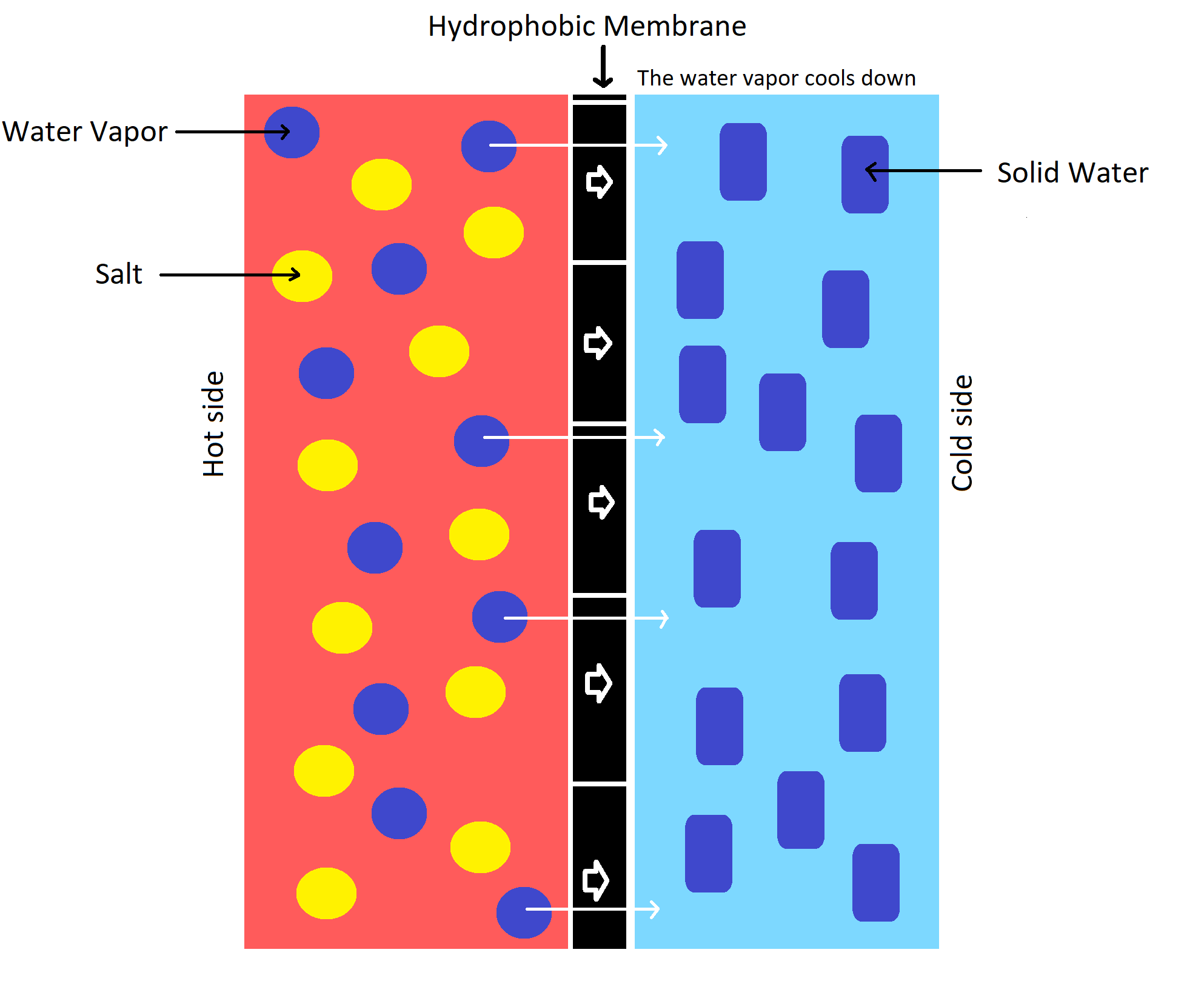
It makes use of a hydrophobic membrane that does not allow water to pass through. However, this membrane does allow vapor to pass through the pores of the membrane. The driving force which allows the vapor to pass through is given by the difference in pressure on both sides of the membrane.
The pores of these membranes are 0.1 and 0.5 µm in diameter width. The difference in pressure is a result of the temperature difference between the two sides of the membrane allowed for the vapor to pass through. The vapor will transport from the hot side of the membrane through it to the colder side.
c) Graphene-Based Sieve
This type of sieve forms part of nanotechnology that enables it to sieve out the salt in seawater and giving clean drinkable water as a result. This type of sieve is best used in combination alongside other methods such as membrane distillation.
Currently, most of the desalination plants across the world, make use of polymer-based membranes. The sieve can also be used alone to filter the salt from the water but will not be as productive as other methods that are used in combination. Graphene is a single layer of carbon atoms that are arranged in a hexagonal shape.
To make this material permeable, you need to drill tiny holes into it that are small enough to block out the salt, but large enough for water molecules to pass through. The holes should also not be larger than one nanometer.
Another problem with this technology is that the sieve becomes swollen when submerged under water. This action will result in the holes becoming smaller and will lower the speed at which desalination will take place. The swelling can be restricted by placing epoxy resin walls on each side.
d) Reverse Osmosis
Reverse Osmosis (RO) is one of the most widespread methods for desalination. It does not make use of temperature but instead uses pressure to push water through a semipermeable membrane.
The salt concentration on the side where the pressure is exercised will increase as the water is pushed through the membrane leaving the salt behind. The membrane can be made out of polyamide film or other polymer-based membranes.
The membranes itself are not that strong and needs additional support to sustain the pressure thrown at it. Energy is required to drive the pumps which will provide the pressure for the saline water to move through the membranes.
The membranes will also need to be continuously cleaned as salt, and other impurities will start to build up. This process can be called flushing where the salt built-up on the membranes are sent back into the ocean which has an adverse effect on the marine life.
e) Vacuum Distillation
This process makes use of heat to turn liquid seawater into boiling water which will cause it to transform into vapor which will leave the salt behind. This method also requires less energy than others that use electricity to boil the water.
Here is why.
It is well known that when atmospheric pressure is reduced that water will start to boil at a lower temperature. This change will result in less heat needed to boil the water which will lead to less dependency on power.
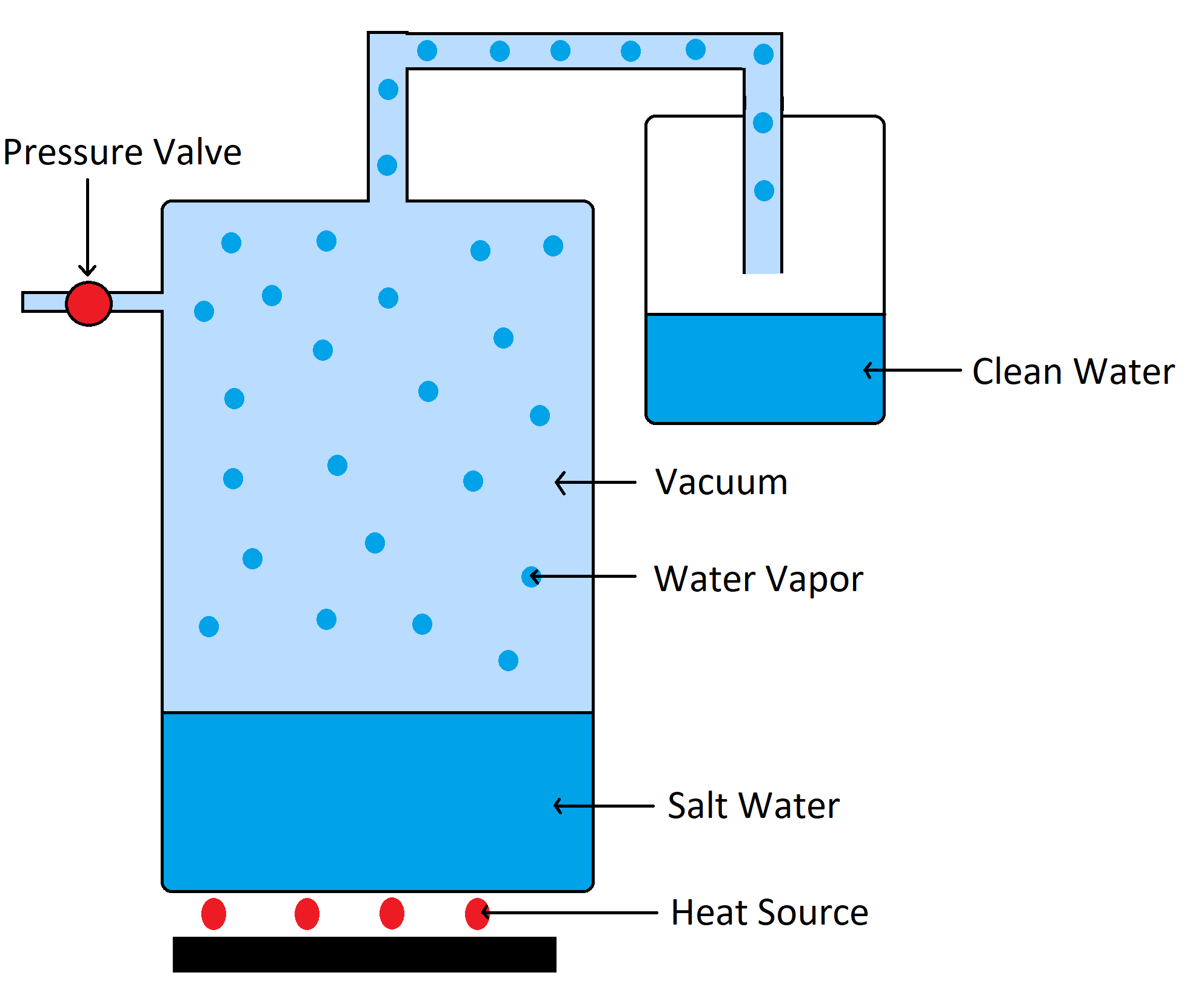
This method is mostly used in tanks that are vacuum induced by sucking out all of the air. The inside pressure of the tanks will lower as a result, and salt water will be prayed inside. The bottom of the tanks will also be heated to produce steam. The steam will then flow through a pipe which will then provide fresh water as the end product.
f) Electrodialysis Reversal
This process makes use of an electrical charge to move salt through charged membranes. Salt has a small electric charge which is drawn out of the water when a current is applied.

The water will be passed through two electrodes which have an opposite charge. The ions in salt have both a positive and a negative charge because Na is positive and Cl is negative.
The positively charged membrane will pull the Cl out of the water, and the negatively charged membrane will pull the Na out of the water which will then leave clean water behind.
This method does not require a lot of energy which saves money and reduces the carbon footprint. You can easily run this system with solar panels because it makes use of a small charge to draw out the salt and other charged impurities.
g) CETO
This method is named after a Greek sea goddess and works alongside reverse osmosis to seperate the salt from water. As already mentioned, reverse osmosis needs pumps to generate pressure for the water to move through the membranes.
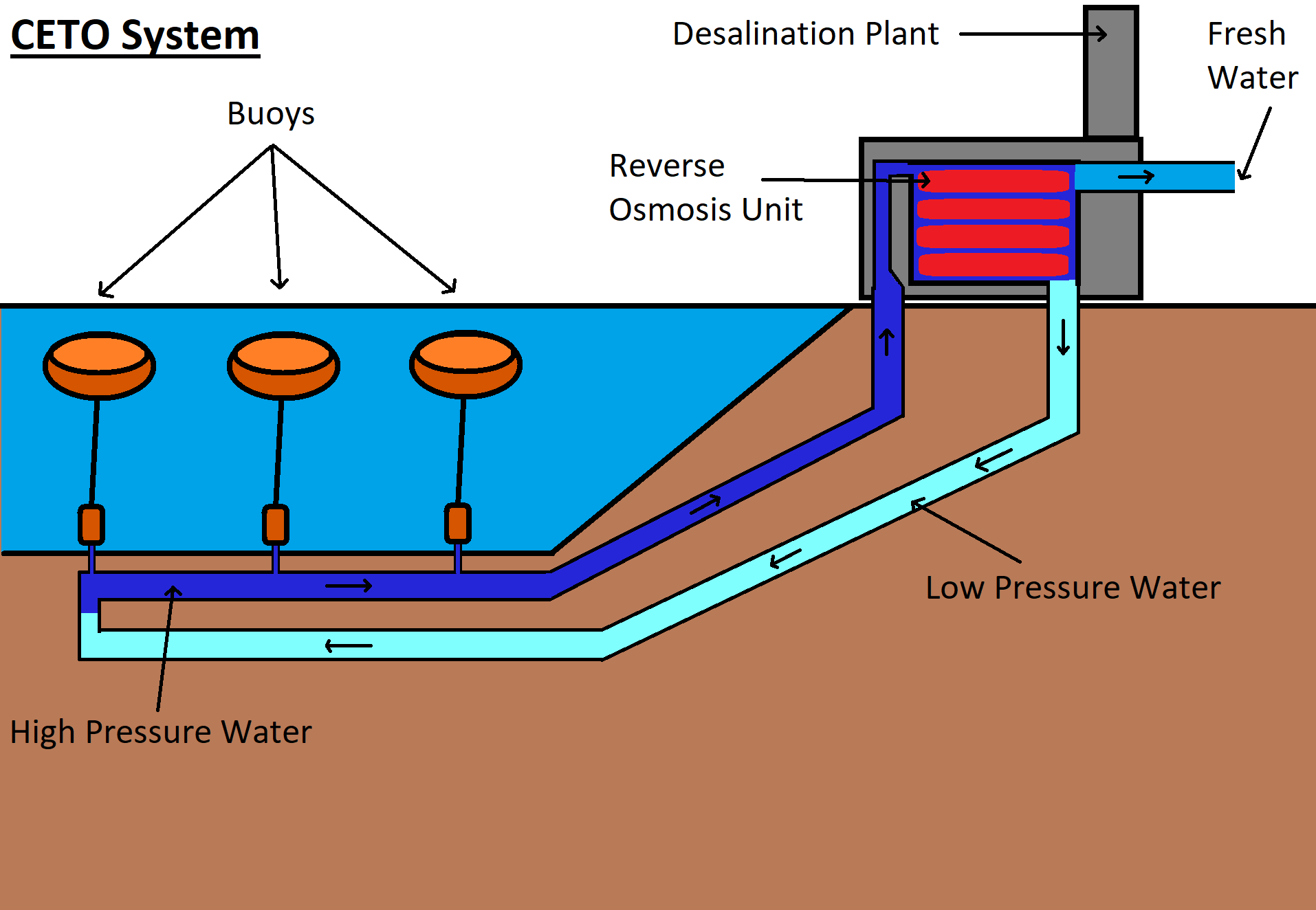
Here is where it gets interesting.
Instead of using pumps that are using electricity, the desalination plants can use the power of Ceto.
What is Ceto?
Ceto is a system that makes use of submerged buoys that will use the ocean swell (underwater waves) to create kinetic energy that can either produce electricity or pressure because of its hydraulic system.
These buoys need to be 20m-50m underwater for it to generate kinetic energy. In the case of desalination, it will automatically pump seawater to the surface at a higher pressure.
This pressure will allow for the water to be pushed through the membranes to produce clean water. This method can reduce the cost associated with the process of desalination where power is involved.
h) Multiple-Effect Distillation
Multiple-Effect Distillation (MED) makes use of multiple stages which turn salt water into fresh water. The seawater is sprayed over hot tubes which then creates steam. Some of the steam is then converted into freshwater while some are being passed onto the next stage.
The steam will then flow to the next stage where it will heat more water which then creates more steam. This creates an effect where each stage will start to reuse the energy in the form of steam that comes from the previous stage.
There is a limit as to how far this process can be taken. The further the stage in the sequence, the more additional energy is required to maintain the heat that is needed to seperate the salt from the water.
i) Freezing-Thaw Desalination
This process relies on the principle that fresh water will freeze before salt water will which in this case will seperate the salt from the clean water. The process works when salt water is sprayed into the air above one of the large pads. The water sprayed into the cold air will then rapidly freeze.
This process needs to happen at sub-freezing temperatures for it to work. A Layer of ice will start to build up until it reaches a specific hight. Over a while, the ice will begin to build-up and will look like a giant ice structure. The surface of the structure will then be cleaned to remove some of the salt and then be melted.The melted water will then have three times less salt than the water that was initially sprayed onto the pad.
j) Vapor-Compression Distillation
The first step in Vapor-Compression Distillation is to heat the salt water until it starts to boil and release vapor in a large closed tank. Once the steam is released above the concentrate, it will be compressed which will increase the pressure of the vapor.
Once the pressure of the vapor starts to increase, the temperature of the vapor will also rise. The same compressed vapor will then be used to provide heat for the concentrate which will produce even more vapor.
This process will save energy in the form of heat which means that you can spend less energy on producing heat to the seawater. You also need to keep in mind that you save more power in the form of the vapor than what you are spending on the compressor.
It will not be productive if you spend more energy to work the compressor than what you are saving with the heat generated by the vapor.
k) Metal-Organic Frameworks (MOFs)
These compounds are made out of metal ions and can have a three-dimensional form. It has a massive internal surface area that makes it ideal to use in the desalination process.

The pores of these compounds are stable enough to filter large molecules from water in this case salt. This compound can be used as a membrane sponge to filter out salt from water. What makes this compound a good option financially is the fact that it also filters out lithium salts which can be used in batteries.
This byproduct can also be sold to large battery manufacturers that can use the lithium inside their batteries.
l) NESMD
Nanophotonics-Enabled Solar Membrane Distillation or NESMD makes use of nanotechnology along with solar panels to transform sunlight into heat. The nanoparticle can capture up to 80% of the sunlight to create enough heat for the distillation process.
This hot water can the easily flow through the membrane and leave the salt behind. What makes this technology different than the other is that it only use sunlight to generate enough heat to start the process. Most of the other methods use a lot of electricity which does not make it cost-efficient.
That is not all.
It can also be used off-grid which means that it is used in rural areas where there is little to none power supply.
How To Clean Water If You Are Stranded
Have you ever imagined what you would do if you found yourself stranded out on the ocean where it stretches as far as the eye can see?
Most people don’t.
Your priority would be to find water. Millions of liters of water surround you, but the only problem is that the water from the sea is not drinkable.
ou can cause more harm to your body than good. What will happen?
Your body will not only consume water but salt as well. Too much salt in the body will cause dehydration at a cellular level. The first thing that your body will try to do is to get rid of the salt.
How exactly?
It will try to get rid of the excess salt through your urine. The more you urinate, the more moisture you will lose. Drinking salt water will be counterproductive at trying to hydrate your body.
a) Use a Plastic Solar Still
If you like to spend a lot of time on the ocean, then you at least need to have a solar still on the boat. A Solar still makes use of the sun’s energy to create enough heat for the salt water to evaporate and then to collect the fresh water.

Most solar stills are inflatable and made out of plastic material that you can pump up like as you would do with a jumping castle. You can either do it with a pump, or you can use your mouth to inject air into the still.
Once it is inflated, you can fill the center with seawater until it is full. The center will also be coated black to absorb more sunlight. The water will start to evaporate and will hit the edges of the still. One enough vapor is formed, it will create water drops which will roll down with the help of gravity.
The water will collect in the circular reservoir located at the bottom of the still around the edges.
b) Cooking the Salt Water
If you are on a boat and you have access to a small gas stove with a pot and a glass, then you are in luck as you can use these items to turn salt water into clean drinkable water.

Here is what you need to do:
- Step 1 – Place the glass in the middle of the pot so that open side of the side is facing upwards.
- Step 2 – Fill the pot with salt water so that it is just below the level of the glass. You do not want the salt water to enter the glass.
- Step 3 – Place the lid upside down on the pot so that the knob is facing downwards. Make sure that the glass is directly under the knob so that the water drops directly into the glass.
- Step 4 – Bring the water to a boil and wait for the glass to fill.
What is going to happen is that the boiled water will release steam that is free from salt. The steam will collect on the surface area of the lid and will start to form droplets. Once the water drops are big enough, it will start to run down the middle of the knob and dip into the glass.
What do you do if you do not have a heat source such as a gas stove?
It is simple.
You use the sun.
Instead of putting the pot on the stove, you can put it outside in the sun so that it follows the same process as that of the solar still.
Conclusion
That’s it for today. I hope you found this article useful and please share with us other useful methods that I might have missed. I hope that we can soon find a long term solution to providing much needed fresh water to humanity.

Leave a Reply